Sharp Center for Research
For Researchers
How to submit a study for review.
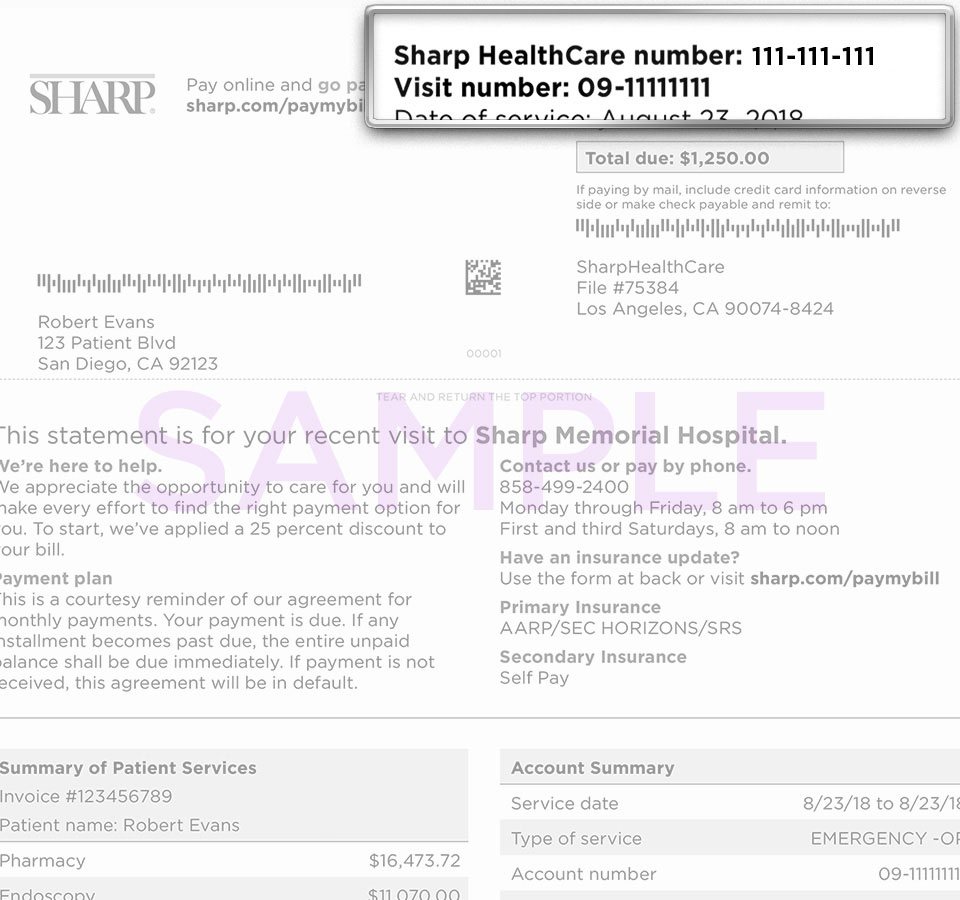
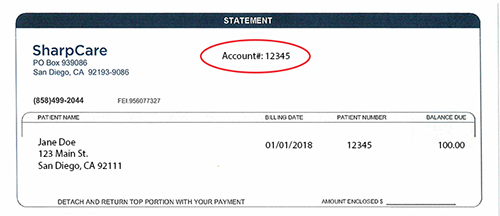
The Sharp HealthCare Institutional Review Board (IRB) reviews research involving human participants that are proposed for Sharp HealthCare entities. The purpose of this review is to protect the participants' safety, satisfy the obligations of Sharp HealthCare and its affiliated entities and to maintain responsible research conduct.
It is the IRB's mission to promote a culture of safety and respect for human beings who choose to participate in research for the greater good of the community.
If you have questions, please contact the IRB administrator at research@sharp.com for guidance regarding the process for submission of new research activities to the Sharp HealthCare IRB. Please remember that no research activity can begin until you receive documentation of the IRB and administrative approvals.
Guidance documents for investigators
You will need Adobe Acrobat Reader to view and download these documents.
HRP-003 Guidance: Research Misconduct
HRP-004 Guidance: Activities that Require IRB Review
HRP-005 Guidance: Outreach and Community Involvement
HRP-006 Guidance: Subject Selection, Recruitment and Payments
HRP-009 Guidance: IRB of Record and Reliance Agreements
HRP-012 Guidance: Observation of the Consent Process
HRP-013 Guidance: Legally Authorized Representatives (Surrogate Consent)
HRP-014 Guidance: Child Assent, Parents or Guardians Permission, Waiver and Special Circumstances
HRP-016 Guidance: Waiver or Alteration to Consent and/or HIPAA Authorization
HRP-022 Guidance: Planned Emergency Research
HRP-023 Guidance: Emergency Use Review
HRP-027 Guidance: Prompt Reporting Requirements
HRP-048 Guidance: Clinicaltrials.gov Registration for SHC-Initiated Clinical Trials
HRP-049 Guidance: Medicare Coverage Analysis
HRP-055 Guidance: Financial Conflicts of Interest
HRP-062 Guidance: Quarterly Investigator Quality Improvement Assessment
HRP-085 Guidance: Genetic Research
HRP-089 Guidance: Humanitarian Use Devices (HUDs)
How to conduct research.
To conduct a research study at Sharp HealthCare, please review the Manual: Investigator Guidance (PDF) or send us an email.